Rate of Occurrence Schizophrenia Study Color and Babies
Abstract
Intervention strategies in adolescents at ultra high-run a risk (UHR) for psychosis are promising for reducing conversion to overt disease, but have just limited impact on functional outcome. Contempo studies suggest that cognition does not further pass up during the UHR stage. As social and cerebral impairments typically develop earlier the first psychotic episode and fifty-fifty years earlier the UHR stage, prevention should also outset much earlier in the groups at chance for schizophrenia and other psychiatric disorders. Early on intervention strategies could aim to better stress resilience, optimize encephalon maturation, and prevent or convalesce agin ecology circumstances. These strategies should urgently be tested for efficacy: the prevalence of ~1% implies that yearly ~22 in every 100,000 people develop overt symptoms of this illness, despite the fact that for many of them—east.g., children with an afflicted kickoff-degree family unit member or carriers of specific genetic variants—increased chance was already identifiable early in life. Our current ability to recognize several adventure groups at an early age not just provides an opportunity, merely besides implies a clinical imperative to act. Time is pressing to investigate preventive interventions in loftier-adventure children to mitigate or forbid the development of schizophrenia and related psychiatric disorders.
Introduction
Current treatment of schizophrenia starts as well late
Schizophrenia is a complex brain disorder with a heterogeneous presentation and variable outcome. Schizophrenia is relatively common, with a prevalence ~1%, depending on gender, country, and degree of urbanicity.1 A substantial proportion of patients with schizophrenia experience marked impairments in multiple domains necessary for daily functioning, affecting their ability to maintain social relationships, sustain employment, and alive independently. In improver, the economic brunt is substantial: In Europe, the cost of schizophrenia-spectrum disorders, including both straight and indirect expenses, was estimated to exist almost 94 billion € in 2010.2 Remission of psychotic symptoms can be achieved for the majority of patients,three,four but social and professional impairments generally persist afterwards remission from psychosis.5,6 The reason is that functional outcome is strongly associated with the presence and severity of cognitive and negative symptoms,7 indicating that positive symptoms (i.e., hallucinations and delusions) are not the cadre symptoms of the illness. Although modest improvements in cognitive and social functioning are doable in adult patients,8 astringent deficits in these domains are hard to overcome. To increase therapeutic impact, we should therefore aim to forestall the evolution of severe social and cognitive impairments before they are established.
Developmental and cognitive abnormalities early in the trajectory of schizophrenia
Overt psychosis is not the kickoff, nor the core feature of schizophrenia, and should consequently non be the principal target for early intervention and prevention. During the past ii decades, research has focused on the menses directly preceding the first psychotic episode when subclinical psychotic features emerge. These ultra high-take chances (UHR) studies consistently observe widespread deficits beyond multiple cognitive domains,9 every bit well equally reduced social abilities10 in youth with attenuated psychotic symptoms. To forbid psychosis in this UHR stage, interventions with psychotherapy and nutritional supplements11,12 have been practical with some success. Yet, current information suggest that the cognitive deficits observed at baseline in UHR individuals do not improve with such interventions and usually remain constant when progressing from the UHR stage to psychosis.13 in fact, most people who see UHR criteria already have a major psychiatric disorder and the majority of those who do non transit to a psychotic disorder still stop up with significant psychopathology and/or social inability.14 Indeed, most of the variance in functional outcome is predicted past neurocognitive decrements already nowadays at the start of the UHR phase, regardless of transition to overt psychosis.fifteen,16
A logical inference from these of import observations is that strategies to preclude social-cognitive deficits should exist practical before in the trajectory, i.e., during childhood and early adolescence, or even pre- or perinatally.17 Several epidemiologic studies demonstrate that children who adult schizophrenia equally adults have, on average, a significantly lower IQ at age iv and 7 years,eighteen compared with commonly developing children. These early babyhood cerebral deficits continue to progress at the onset of adolescence, with reduced cognitive performance at historic period 12 years,19 as well as significantly lower school performance at 13 and 14 years.20 Chiefly, a meta-assay on IQ during the course of schizophrenia indicates further cognitive turn down occurring earlier the onset of boyhood.21 This was confirmed in a longitudinal birth cohort with a lengthy follow-upward flow observing that children, subsequently diagnosed with schizophrenia in machismo, had a cognitive decline of nine IQ points at historic period thirteen and a decline of fifteen IQ points around the time of diagnosis as compared with peers who did non develop the disorder.22 Recently, findings in a large prospective longitudinal cohort of patients at increased risk for schizophrenia due to the 22q11.2 deletion showed a similar decrease in IQ, preceding the first psychotic episode by 7 years on average.23 Children who develop schizophrenia may face other problems, east.g., with motor coordination and behavior.24
From these observations, we can infer that several different neurodevelopmental pathways may atomic number 82 to schizophrenia, while similar neurodevelopmental deviations may pb to different psychiatric disorders. Therefore, preventive interventions for schizophrenia are likely to target different risk groups and may subtract risk not only for schizophrenia and related psychotic disorders, but too for a broader range of mental disorders, including affective, personality, and substance corruption disorders.25
The goal of this paper is to discuss early interventions that may have the potential to improve outcome by safeguarding cerebral and social development (primary goal) or by preventing the total-diddled manifestation of psychosis (secondary goal). We will besides outline which specific subgroups within the population may be nigh amenable to such strategies and, finally, what upstanding and economic aspects are relevant in relation to the utilize of very early on prevention strategies.
How to arbitrate: potential strategies for prevention in at-risk groups
Beginning, we suggest strategies that can ameliorate suboptimal maturation of neuronal pathways during childhood. Second, we examine interventions that tin can reduce environmental insults or mitigate their touch. Third, we explore strategies that can improve resilience, even in the presence of negative circumstances and/or genetic risk factors.
Improving suboptimal maturation of neuronal pathways
While aberrant dopaminergic signaling is strongly associated with the onset of psychosis,26 gamma-aminobutyric acrid (GABA)-ergic and glutamatergic signaling defects may be critically involved in the evolution of social and cognitive deficits in schizophrenia.27–29 While GABA is an inhibitory neurotransmitter in adult life, it is excitatory in early fetal brain evolution. The chloride transporter KCC2 switches GABA from excitatory to inhibitory. This chloride transporter can be stimulated by activation of postsynaptic α7-nicotinic acetylcholine receptors.30
Glutamatergic compounds
To date, several strategies to alter glutamatergic neurotransmission have been investigated for their effect on noesis in patients with schizophrenia using glycine-site NMDA-modulating compounds such as glycine, d-serine, d-cycloserine, and the glycine transporter one (GlyT1) inhibitor sarcosin.31 The bear witness with regard to the addition of the NMDA receptor partial agonist d-cycloserine to antipsychotic therapy is mixed, with either exacerbation or alleviation of positive symptoms32 and amelioration of negative symptoms.33 Efficacy of d-serine treatment with regard to cognitive symptoms in schizophrenia has non been convincingly demonstrated.34,35 In addition, a recent meta-analysis demonstrated that glutamate-positive modulators are not benign in the treatment of cognitive symptoms in schizophrenia.36 Hence, studies thus far have not shown efficacy of these glutamate-modifying compounds in adult patients with schizophrenia. All the same, this arroyo claim further exploration, in item the utilize of such agents during the early phases of schizophrenia. If practical early, when abnormalities in glutamatergic manual start to emerge in those who develop schizophrenia, their efficacy may be increased. Indeed, a recent preclinical study showed far-reaching normalizing effects of d-serine on brain function in Pick1 knockout mice, provided it was applied early. The behavioral deficits associated with loss of part of Pick1 could be reversed, merely but when d-serine was administered neonatally, not during adult age.37 In addition, there may be other glutamatergic-modulating agents with potential. For instance, there is emerging evidence for a therapeutic effect of pregnenolone in patients with schizophrenia, a molecule with both neurosteroid and NMDA receptor-modulating effects.38 More clinical studies are required to examine effects and possible adverse effects of these and other glutamate-modifying drugs, while pre-clinical studies are needed to elucidate the mechanisms of activity of these molecules on diffusive neurodevelopmental pathways and the optimal timing of such interventions.
GABA-ergic compounds
Some other potential arroyo to enhancing cognition in those at risk for psychosis is by influencing GABAergic pathways. Two important classes of selective GABAergic drugs show promise: αfive-selective inverse GABAA receptor agonists and α2/3-selective GABAA receptor agonists.39 The αv subunit, which is predominantly expressed in the hippocampus, is essential in modulating the interneuron-pyramidal network. While the evidence for pro-cerebral effects of αv-selective inverse agonists is equivocal in humans,40 at that place is potential for modulation of GABAA receptors with an α5 subunit.41,42 The results of a first study on pro-cognitive effects in adult patients with schizophrenia, using the αii/3-selective positive allosteric modulator MK0777, were disappointing.42 Yet, the rationale for this strategy remains appealing given the large body of evidence for altered expression and functionality of GABAA receptor αtwo/3 subunits in schizophrenia and their relevance for cognition.39 In particular, similar to the glutamate system, it is possible that GABAergic interventions are more than effective when applied earlier in life, when the GABA system is notwithstanding building up neural connections and therefore more acquiescent to change. In addition to modifying GABAA receptor functionality, other possible approaches to attune the GABA system should too be studied, including modulating GABA metabolism, using cation/chloride transporters, neurosteroids or transcranial magnetic stimulation.43 For example, bumetanide, which inhibits the cation/chloride transporter NKKC1 and thereby indirectly modulates GABAergic transmission, has been shown to change cortical circuits and reverse sensorimotor-gating deficits.44,45 Nonetheless, interference with the GABAA receptors is not without danger, given the increased hazard for triggering epilepsy. Therefore, the development of alternative approaches to modify GABAergic neurotransmission with milder side-effect profiles is needed.
Choline supplementation
Perinatal supplementation with choline is shown to ameliorate cognitive function in animal models of schizophrenia.46 A trial in 100 homo newborns showed that babies with perinatal supplementation more ofttimes showed acceptable inhibition at the auditory P50 paradigm than infants without supplementation,47 indicating that a choline-enhancing diet for significant women with high risk or supplementation for neonates may accept positive effects on cholinergic part and facilitate the evolution of adequate cortical inhibition.
Anti-oxidants and agents that affect the allowed response
Findings from murine studies suggest that both glutamatergic and GABA-ergic manual may be negatively affected past increased immune activation and a negative redox residuum in the encephalon.48 A negative redox remainder causes oxidative stress, and results from an abundance of reactive oxygen and nitrogen species relative to the availability of antioxidants. Both immune system and redox abnormalities have been observed in the peripheral blood of patients with schizophrenia, but also in those at increased run a risk.49–52 The nearly important antioxidant for the brain is glutathione, which plays a critical role in myelination and white thing maturation,53 and can be replenished by nutritional supplement of its amino acrid forerunner N-acetylcysteine (NAC). NAC is not-toxic, has few side effects54 and induces an upregulation of glutathione synthesis, which can neutralize extra production of oxygen and nitric radicals in a stressed brain.48 NAC also has mild anti-inflammatory effects, likely via its antioxidant properties.54 Several rodent studies demonstrate that restoration of the redox residue by NAC or other antioxidants during early on developmental stages tin can mitigate adverse effects of stress on brain maturation,55 and rescue social or cognitive deficit phenotypes induced past early social isolation,56 or preclude the development of these phenotypes induced past neonatal hippocampal lesions.57 In add-on to replenishing glutathione, NAC also plays a role in the regulation of synaptic NMDA signaling.58 Its positive effects on the redox balance, neuroinflammation, and NMDA receptor functioning in combination with its mild side-effect profile make NAC a preferred candidate molecule for further study with preventive potential when applied early in the grade of schizophrenia.
Omega-iii type polyunsaturated fatty acids (PUFAs) too have some antioxidative capacity59 and balmy anti-inflammatory effects on the brain.60 PUFAs are an important component of neuronal and glial cell membranes and could facilitate synaptogenesis. Like NAC, PUFAs have a very mild side-upshot contour, and thus would be a good candidate to study preventive furnishings when used early in development. One report reported significant positive effects of early on administration of PUFAs compared with placebo in youth at hazard for psychosis on both the transition rate to full-diddled psychosis and broader functional effect measures.61 Currently, recruitment for a new written report is ongoing to replicate this finding.62 PUFAs were shown to accept some benign effects on neurodevelopment and could mitigate the risk for babyhood psychiatric disorders such equally autism and ADHD.63 As all neurodevelopmental disorders form gamble factors for schizophrenia,64,65 this could be an additional route to subtract the risk for schizophrenia in at-risk groups.
Another potential intervention to attune the immune response with minimal side furnishings is the use of probiotics. The large impact of the microbiome on brain function is becoming increasingly clear. Several gut bacteria are capable of producing neurotransmitters such every bit GABA and acetylcholine.66 A "leaky gut" has been hypothesized to pb to increased inflammatory activation and have negative impact on encephalon maturation in several psychiatric disorders, including schizophrenia.67 Probiotics contain beneficial bacteria such as bifidobacterium and lactobacillus that can subtract systemic proinflammatory cytokines, increase neurotrophic factors, and reduce oxidative stress.68 In good for you adults, these agents have been shown to reduce feet and stress.69 Since probiotics have few side effects, this may exist a promising intervention for children at increased adventure to develop schizophrenia.
Reducing environmental insults or their touch on
The fundamental biological organization required for stress accommodation, the hypothalamic-pituitary-adrenal (HPA) axis, is abnormal in schizophrenia and altered HPA axis functionality has been related to both cerebral and negative symptoms in schizophrenia.70–72 Moreover, changes in the HPA axis may predate the onset of overt psychotic symptoms since UHR patients brandish a blunted cortisol stress response,73 altered basal cortisol levels,74 and abnormal pituitary volumes.75 In improver, the entreatment of interventions aiming to reduce stress and improving resilience is based on strong evidence linking excessive stress with biological mechanisms associated with schizophrenia, including key nervous system immune activation,76 dopamine77 and glutamate76 release, and redox remainder disruption.78 In addition to interventions directly aimed at reducing social stress, the prevention of drug abuse during adolescence may also be a relevant strategy given the bidirectional relation between drug abuse and stress. Not only are the exposure to excessive stressors and alterations in the HPA centrality risk factors for drug corruption,79 the use of drugs can as well be associated with a disruption of normal stress regulation.80
Social skills training to prevent bullying and social exclusion
Reducing bullying and peer rejection may improve social outcomes in the general population.81 Targeted anti-bullying programs therefore found a promising prevention strategy, equally bullying is frequent among youth predisposed to schizophrenia or related disorders and often results in social isolation and chronic stress.82,83 The use of an private coaching program for children and young adolescents can decrease the prevalence of bullying and meliorate social and cognitive functioning.84 Children who already have some delay in cognitive, social, or motor evolution are at even higher risk for beingness bullied and if that happens their risk for many different psychiatric disorders increases.85 A prospective written report on >ane,000 adolescents showed that the incidence of psychotic experiences decreased significantly in individuals whose exposure to bullying ceased over the grade of the report,86 indicating that interventions that tin finish bullying can touch the expression of psychosis vulnerability. These findings underscore the relevance of such programs in reducing stress from bullying and social isolation; the time has come therefore to examine their preventive potential when applied at an early age in children who are at increased risk for schizophrenia.
Early on interventions to prevent drug corruption
In improver to its relation to stress regulation, drug abuse, particularly when initiated in the early teens, has long been recognized as an of import take a chance cistron for schizophrenia.87 Although more studies are required for conclusive show,88 results thus far betoken that interventions for teens and their parents, which ameliorate family communication and rule-setting, can reduce the rate of subsequent drug abuse and accompanying problem behavior.88,89 Given the serious consequences of early on-drug use in youth at run a risk for schizophrenia, studying the efficacy of such programs in these populations is urgent.
Improving resilience
Cognitive remediation
The main goal of cognitive remediation (CR) is to improve neuropsychological deficits, making it an appealing strategy to examine in the very early stages of schizophrenia in high-adventure subjects, when cerebral deficits emerge. To appointment, there is increasing bear witness for the benefits of CR applied after the first psychotic episode in patients with schizophrenia, with medium upshot sizes on global cognition, which remain measurable afterwards at least 1 year.ninety Interestingly, these effects are larger when applied at younger age,91 after a shorter duration of the illness,92 and in individuals with higher cognitive levels.93 Benefits of CR practical to individuals in the UHR period with subclinical psychotic symptoms have been examined, indicating both cognitive and social gains.94 CR may be more constructive when given in the period earlier the UHR stage when cognition can still be saved, i.e., during childhood.
Exercise training
Concrete exercise may improve performance on different cerebral measures in patients with schizophrenia. Interestingly, exercise is associated with changes in factor expression related to brain plasticity,95 and improvements in both brain structure96 and connectivity.97 Although the majority of studies investigating the benefits of practise take focused on the elderly, there is some evidence for similar efficacy in youth,98 making it an fantabulous intervention for master or secondary prevention of cognitive decline and the development of severe psychiatric disorders.
Who to target: potential candidates for early-preventive strategies
When the focus of potential intervention is shifted from the UHR phase to an earlier phase of development, more than subjects will be exposed to interventions, thus necessitating an even more than rigorous consideration of potential negative furnishings of at-risk designation and intervention strategies. Selected populations with high odds ratios to develop schizophrenia may provide a rational starting point for such studies, especially since fifty-fifty children from these take chances groups who will non continue to develop schizophrenia may still be affected past cognitive and social impairments.
Children with first-degree relatives with schizophrenia
Children with first-degree relatives suffering from schizophrenia-spectrum disorder have, on average, a 10-fold increased run a risk to develop the disease themselves.99 Cognition is also affected in adult first-degree relatives—even in those who have not themselves developed schizophrenia—albeit to a lesser degree compared with the impairments observed in clinically affected individuals.ix Hazard for schizophrenia is not only increased in children from schizophrenia patients, but besides in children from those with bipolar disorder.100 Interventions to prevent the development of cerebral deficits may therefore be relevant to those youngsters who are in fact early in the trajectory of schizophrenia, but too for the rest of this group who will not develop the disease.
Children with 22q11.ii deletion syndrome
The 22q11.2 deletion syndrome (22q11DS) has an estimated incidence of 1 in 2,000 and is the strongest unmarried genetic risk factor for schizophrenia currently known, with approximately one in 4 individuals with 22q11DS developing the disease. In these individuals, the principal clinical characteristics cannot be distinguished from schizophrenia in the general population.101 Notably, a substantial proportion of 22q11DS children, when followed prospectively, display a decrease in IQ over time.102 Moreover, consequent with the observed cerebral abnormalities predating the onset of psychosis in the full general population (see Figure ane),19,20 22q11DS children who showed an early-cognitive pass up had a threefold increased risk to be diagnosed later on with a schizophrenia-spectrum disorder, compared with those without cerebral pass up.23 The observation of increased plasma levels of the amino acrid proline in approximately one 3rd of individuals with 22q11DS103 is notable as evidence suggests that proline influences glutamatergic neurotransmission.104,105 Indeed, findings of several studies indicate that high plasma proline levels influence brain function in 22q11DS patients,106,107 suggesting that strategies to alter glutamatergic neurotransmission may be of particular relevance for this population. In conclusion, given their relative loftier-conversion rate and well-documented cognitive decline, children with a known 22q11 deletion are a small, but appealing target population for early on-intervention strategies.
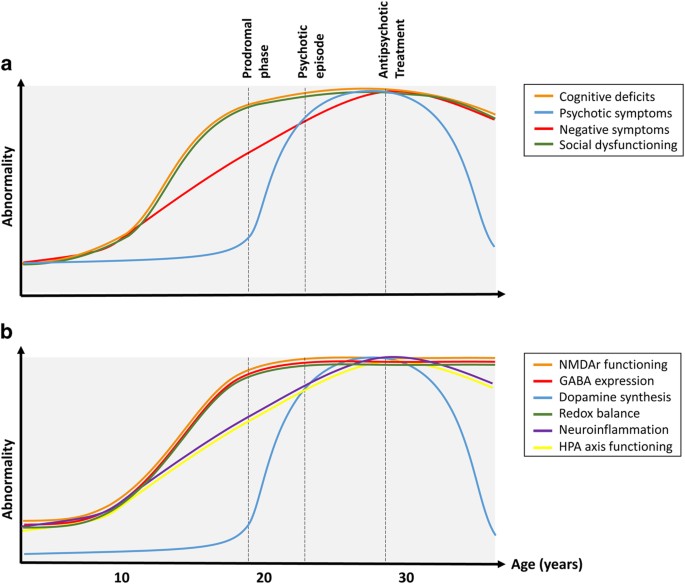
Hypothesized typical course of schizophrenia. (a) shows the clinical class of the disease. (b) shows the hypothesized course of the underlying molecular mechanisms.
Carriers of other CNVs associated with schizophrenia
In addition to 22q11DS, other copy number variants (CNVs) are besides associated with increased risk of schizophrenia, including microdeletions at 1q21.1, 3q29, 15q11.2, and 17q12, every bit well as duplications at 15q11-13, 15q13.3, 16p11.2, and 16p13.one.108 Each of these variants is also associated with increased rates of other neurodevelopmental phenotypes, particularly autism spectrum disorders, intellectual disability, and epilepsy,109 also as aberrant morphological features. These characteristics may cause this risk group to be vulnerable to social stress, therefore strategies to amend resilience and reduce social stress may be of item benefit. These CNVs occur at very low rates in the population108 and even when considered together, they include only a small-scale proportion of all schizophrenia patients. In dissimilarity, the risk to develop schizophrenia in individual carriers of whatever one of these CNVs is substantial, with estimated odds ratios ranging betwixt 2 and >thirty,108 making the population of carriers of these CNVs a target group for utilise of preventive measures early in the developmental trajectory.
Youth experiencing transient psychotic symptoms
Psychotic experiences are mutual amidst young children, occurring in well-nigh xv% of children aged ix–11 years old.110 In about cases, these symptoms are transient, but approximately ane third of this grouping experiences psychotic symptoms for over a year. For children with psychotic symptoms, the adventure to develop a schizophrenia-spectrum disorder by the age of 26 is between 5 and 16 times increased, depending on the number and severity of the psychotic experiences.111
Children with psychotic experiences are not only at increased risk for schizophrenia, but likewise for other psychiatric disorders,112 and therefore general interventions to forbid deterioration may be of added value. Based on retrospective information obtained from adult patients with schizophrenia, 40% already experienced one or more psychotic features in childhood.fifteen Thus, preventive measures should be considered in children with psychotic experiences, in particular when they are persistent, when more than one feature is present, or when one severe psychotic symptom is nowadays.
Ethical and economical aspects of early on-preventive strategies
With the exception of the pharmacological agents, the interventions discussed under 'How to intervene: potential strategies for prevention in at-gamble groups' are non-invasive and prophylactic, thereby fully respecting the adagium primum non nocere (i.due east., first do no harm). Furthermore, some interventions, such as do training and bullying prevention programs are not-specific and likely to be benign for any youth, regardless the risk of developing schizophrenia. Nevertheless, the identification as a person at risk may accept negative bear on on a person'due south well-existence. Furthermore, without exception, the suggested interventions volition exist costly and it is therefore a cardinal to provide the intervention for those who need information technology most.
Disclosing presence of psychotic experiences and at-risk status
When communicating information about at-run a risk status for astringent disorders to children and their families, a leading ethical question is whether telling children well-nigh their at-risk status harms them. This question should be considered in the context of the anticipated gain of the intervention, which is the prevention or mitigation of social and cognitive impairments and, ideally, of the total manifestation of a major psychiatric disorder. Communicating information about an increased risk for a major psychiatric disorder may cause anxiety, merely it may likewise be beneficial for both the child and the family, as it may validate perceived social or cognitive problems or emerging psychotic experiences. Disclosure also provides the opportunity to educate child and family, for instance about other potential take a chance environments, early-symptom recognition, and lifestyle adjustments. More all-encompassing discussions of considerations regarding upstanding aspects of disclosing risk status take recently been published.113,114
Economical aspects of early-intervention strategies
Even when focusing on selected groups with high a priori risk for schizophrenia, it is important to assess the economical feasibility of early on-intervention strategies. What are the expenses required for the implementation of the interventions and how do they weigh against the expected gains, i.e., the number of patients in whom a cerebral and social turn down, and full development of schizophrenia is averted and improvement in cognitive and social functioning in those who practise not make a transition to a psychotic disorder? In the absence of reliable data on issue sizes of the interventions discussed in this paper, we have modeled several scenarios to provide some insight into this question. In Effigy 2, nosotros present three scenarios (Figure 2a–c) based on unlike outcome size assumptions, which are expressed equally the prevention rates of the intervention (5%, 10%, and 25% respectively). For instance, a prevention rate of 10% implies that in 1 out of 10 individuals who are exposed to the intervention, the onset of social and cerebral dysfunction, and full development of schizophrenia is prevented. The general population, with a lifetime incidence of about 1%, is provided as a reference. The figure should be viewed while keeping in mind a conservative estimate of the economic burden related to ane individual who develops a psychotic disorder, i.e., ~167k € per 10 years (indicated with the dotted blackness line).115 While interventions at low costs may be toll-effective in high-risk groups, we should also consider that there is a general willingness to pay for preventive interventions to safeguard futurity generations from astringent dysfunction.
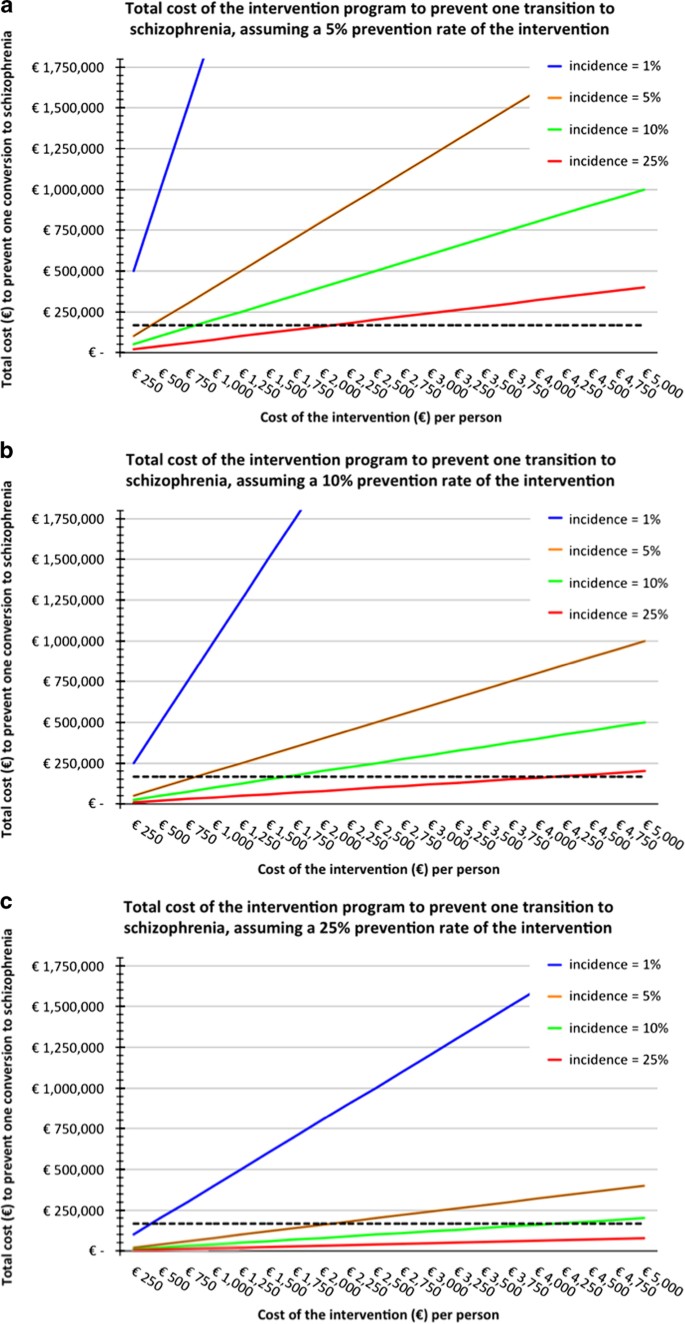
(a–c) Economical feasibility of early-intervention strategies. This figure indicates the total cost (in €) of any intervention strategy (ten axis) to prevent the development of schizophrenia in one private (y axis). (a–c) represent three scenarios based on different prevention rates of the intervention (5%, 10%, and 25% respectively). For reference, the black dotted line represents the estimated economic burden of ane individual who develops a psychotic disorder calculated for a time span of 10 years. The area under the dotted line indicates an economical do good since the costs of the preventive strategy to prevent ane transition to schizophrenia outweighs the economic burden of one affected private. Several factors influence whether a preventive intervention is cost-effective, including (i) the costs of the intervention (x axis). (2) lifetime risk for a psychotic disorder in the target population (different line colors) with lower risks resulting in a reduced toll-effectiveness. The blue line refers to the risk for schizophrenia in the general population (1%), the red line applies to the selected population of 22q11DS individuals with a 25% lifetime incidence of schizophrenia and (three) the effectiveness of the intervention; that is, in what proportion of individuals can the evolution of a psychotic disorder exist averted as a consequence of the intervention?
It is likewise important to realize that all the aforementioned take chances groups have increased odds of multiple psychiatric disorders, non just schizophrenia. Therefore, early on intervention in any of these groups may as well be relevant in the prevention of a broad spectrum of psychiatric disorders, thereby enlarging the predictable gain of the intervention. Finally, the selection of a high-risk population can be farther improved when more expensive interventions are to exist examined. This could be accomplished by selecting those children who accept more ane hazard factor. For example, youth with subclinical psychotic symptoms and a family unit history of schizophrenia may be a strategic population to written report such interventions before whatsoever associated cognitive dysfunction progresses to more severe levels.
Give-and-take and conclusions
The by 20 years have largely increased our agreement of the developmental trajectory of schizophrenia. We now take some basic cognition on environmental circumstances and neurobiological factors that tin can positively or negatively affect this developmental trajectory. Yet, this cognition is not translated into preventive interventions in early stages. Each decade, many young people around the world develop schizophrenia without even a trend towards lowering the incidence. Time is pressing for research into early-preventive interventions to diminish the development of schizophrenia-spectrum disorders in futurity generations.
Psychosis is a core, only also a late symptom of schizophrenia, generally starting in early adolescence during the UHR catamenia and progressing to the first psychotic episode in immature adulthood. However, cognitive and social dysfunctions emerge at a much earlier stage and evolve to become the virtually disabling symptoms. Compromised cerebral performance in children who subsequently develop schizophrenia as compared with their peers is already nowadays at age 4116 and may well be impaired from the very get-go. In addition, in that location is bear witness for a deterioration in cognitive and social operation in early adolescence.21 While interventions to save psychotic symptoms in the UHR flow or during psychotic episodes have only limited impact on long-term functional consequence, preventing cognitive and social decline during childhood or early on adolescence may have substantial impact on long-term functioning. In this perspective, we advise that the term premorbid may no longer be accurate when used in schizophrenia equally it erroneously suggests that the disease process starts with either the prodromal stage or the onset of the first psychotic symptoms.
Several interventions that are well-tolerated tin can potentially prevent astringent cognitive dysfunction when applied early in the form of the disorder. Such interventions may aim to avoid or mitigate disadvantageous circumstances, such equally bullying, social exclusion, or drug use in children at increased take chances. Other strategies include the optimization of brain maturation or resilience with interventions to ameliorate the redox rest or immune status of the brain. Since abnormalities in the development and maturation of NMDA and GABA receptors may play an of import role in the decline of cognition during childhood and adolescence, this also provides a potential mechanistic background for preventive interventions.
Given the population rate of schizophrenia of about i%, unfeasibly large samples would be required to provide sufficient power for the study of the efficacy of the interventions discussed here. However, we can at present identify a substantial number of selected run a risk populations in childhood on the basis of genetic or clinical characteristics, such as those with first-degree relatives with schizophrenia, with known genome abnormalities, or children experiencing one or more psychotic symptoms. These early identifiable risk groups provide a rational starting betoken to examine the efficacy of existing and promising early on interventions, with regard to the prevention of social and cognitive deficits in schizophrenia.
Moreover, it is plausible that in the near future neuroimaging,117 electroencephalogram, or blood-based biomarkers can be used to further improve the choice of individuals. For instance, reduced P300 aamplitude may already be present during babyhood and could thus be used to improve selection.118 Another case is the measurement of increased inflammatory parameters (due east.g., C-reactive protein, IL1β, IL-half dozen, or other parameters) in children with psychotic symptoms to prioritize those who may do good near from supplements such as NAC or PUFA.
Although the field generally agrees that early on intervention is i of the most important goals in schizophrenia research, just few studies directly test efficacy of such intervention strategies. A possible explanation for this paucity is that most hazard groups take rather depression conversion rates, with only 10–25% of youngsters developing the disorder. This may indeed be a reason for not testing interventions with moderate to severe side effects. Yet, most preventive interventions described in this manuscript accept very mild side-effect profiles, and even accomplish across not nocere as they are beneficial even to those who will not develop schizophrenia.
Nosotros now have a basic understanding of how to human activity early on and therefore it is time to proceed. Randomized clinical trials are urgently needed to provide a scientifically sound footing for the utilize of early-intervention strategies in clinical exercise. Moreover, nosotros demand to evaluate the cost-effectiveness of these interventions. The loftier-adventure groups discussed in this review provide an appealing and strategic starting bespeak. The required trials will exist plush and challenging merely information technology is time they are initiated, given their high potential to uncover novel possibilities for preventing or at least mitigating the course of schizophrenia, peradventure even in the virtually future.
References
-
McGrath, J., Saha, Due south., Chant, D. & Welham, J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol. Rev. 30, 67–76 (2008).
-
Gustavsson, A. et al. Price of disorders of the brain in Europe 2010. Eur. Neuropsychopharmacol. 21, 718–779 (2011).
-
Buchanan, R. W. et al. The 2009 schizophrenia PORT psychopharmacological treatment recommendations and summary statements. Schizophr. Bull. 36, 71–93 (2010).
-
Dixon, L. B. et al. The 2009 schizophrenia PORT psychosocial treatment recommendations and summary statements. Schizophr. Bull. 36, 48–70 (2010).
-
Harvey, P. D. Disability in schizophrenia: contributing factors and validated assessments. J. Clin. Psychiatry 75 Suppl 1, fifteen–20 (2014).
-
Kahn, R. Southward. & Keefe, R. South. Schizophrenia is a cognitive illness: time for a change in focus. JAMA Psychiatry 70, 1107–1112 (2013).
-
Lepage, M., Bodnar, M. & Bowie, C. R. Neurocognition: clinical and functional outcomes in schizophrenia. Can. J. Psychiatry 59, 5–12 (2014).
-
Mueller, D. R., Schmidt, S. J. & Roder, V. I-yr randomized controlled trial and follow-upwardly of integrated neurocognitive therapy for schizophrenia outpatients. Schizophr. Balderdash. 41, 604–616 (2015).
-
Bora, E. et al. Cognitive deficits in youth with familial and clinical high risk to psychosis: a systematic review and meta-analysis. Acta Psychiatr. Scand. 130, 1–15 (2014).
-
Addington, J. & Heinssen, R. Prediction and prevention of psychosis in youth at clinical high risk. Annu. Rev. Clin. Psychol. viii, 269–289 (2012).
-
Ising, H. Yard. et al. Toll-effectiveness of preventing first-episode psychosis in ultra-high-risk subjects: multi-centre randomized controlled trial. Psychol. Med. 45, 1435–1446 (2015).
-
Preti, A. & Cella, Thou. Randomized-controlled trials in people at ultra high hazard of psychosis: a review of treatment effectiveness. Schizophr. Res. 123, 30–36 (2010).
-
Bora, E. & Murray, R. Yard. Meta-analysis of cognitive deficits in ultra-loftier run a risk to psychosis and beginning-episode psychosis: do the cognitive deficits progress over, or after, the onset of psychosis? Schizophr. Bull. 40, 744–755 (2014).
-
Lim, J. et al. Affect of psychiatric comorbidity in individuals at Ultra High Risk of psychosis—Findings from the Longitudinal Youth at Risk Study (LYRIKS). Schizophr. Res. 164, viii–14 (2015).
-
Lin, A. et al. Neurocognitive predictors of functional outcome two to 13 years after identification as ultra-high risk for psychosis. Schizophr. Res. 132, 1–7 (2011).
-
Niendam, T. A. et al. The course of neurocognition and social functioning in individuals at ultra high risk for psychosis. Schizophr. Bull. 33, 772–781 (2007).
-
Arango, C., Fraguas, D. & Parellada, M. Differential neurodevelopmental trajectories in patients with early-onset bipolar and schizophrenia disorders. Schizophr. Bull. twoscore Suppl 2, S138–S146 (2014).
-
Agnew-Blais, J. C. et al. Early childhood IQ trajectories in individuals later developing schizophrenia and affective psychoses in the new england family studies. Schizophr. Bull. 41, 817–823 (2015).
-
van Oel, C. J., Sitskoorn, M. Thousand., Cremer, Chiliad. P. & Kahn, R. S. Schoolhouse functioning as a premorbid marker for schizophrenia: a twin study. Schizophr. Balderdash. 28, 401–414 (2002).
-
Ullman, Five. Z., Levine, S. Z., Reichenberg, A. & Rabinowitz, J. Real-world premorbid functioning in schizophrenia and affective disorders during the early teenage years: a population-based report of school grades and instructor ratings. Schizophr. Res. 136, 13–18 (2012).
-
Woodberry, 1000. A., Giuliano, A. J. & Seidman, L. J. Premorbid IQ in schizophrenia: a meta-analytic review. Am. J. Psychiatry 165, 579–587 (2008).
-
Meier, M. H. et al. Neuropsychological reject in schizophrenia from the premorbid to the postonset period: evidence from a population-representative longitudinal report. Am. J. Psychiatry 171, 91–101 (2014).
-
Vorstman, J. A. et al. Cognitive refuse preceding the onset of psychosis in patients with 22q11.2 deletion syndrome. JAMA Psychiatry 72, 377–385 (2015).
-
Liu, C. H., Keshavan, G. Due south., Tronick, E. & Seidman, L. J. Perinatal risks and babyhood premorbid indicators of later psychosis: next steps for early on psychosocial interventions. Schizophr. Bull. 41, 801–816 (2015).
-
Seidman, L. J. & Nordentoft, Chiliad. New targets for prevention of schizophrenia: is it time for interventions in the premorbid phase? Schizophr. Balderdash. 41, 795–800 (2015).
-
Howes, O. D. & Kapur, Southward. The dopamine hypothesis of schizophrenia: version III--the final common pathway. Schizophr. Bull. 35, 549–562 (2009).
-
Kahn, R. S. & Sommer, I. E. The neurobiology and handling of offset-episode schizophrenia. Mol. Psychiatry 20, 84–97 (2014).
-
Schmidt, M. J. & Mirnics, Chiliad. Neurodevelopment, GABA arrangement dysfunction, and schizophrenia. Neuropsychopharmacology 40, 190–206 (2015).
-
Pocklington, A. J. et al. Novel findings from CNVs implicate inhibitory and excitatory signaling complexes in schizophrenia. Neuron 86, 1203–1214 (2015).
-
Liu, Z., Neff, R. A. & Berg, D. 1000. Sequential coaction of nicotinic and GABAergic signaling guides neuronal development. Science 314, 1610–1613 (2006).
-
Tsai, G. Eastward. & Lin, P. Y. Strategies to heighten N-methyl-D-aspartate receptor-mediated neurotransmission in schizophrenia, a critical review and meta-analysis. Curr. Pharm. Des. sixteen, 522–537 (2010).
-
van Berckel, B. N. et al. D-cycloserine increases positive symptoms in chronic schizophrenic patients when administered in addition to antipsychotics: a double-blind, parallel, placebo-controlled written report. Neuropsychopharmacology 21, 203–210 (1999).
-
Goff, D. C. et al. A placebo-controlled trial of D-cycloserine added to conventional neuroleptics in patients with schizophrenia. Curvation Gen Psychiatry 56, 21–27 (1999).
-
Weiser, M. et al. A multicenter, improver randomized controlled trial of low-dose d-serine for negative and cognitive symptoms of schizophrenia. J. Clin. Psychiatry 73, e728–e734 (2012).
-
Kantrowitz, J. T. et al. High dose D-serine in the treatment of schizophrenia. Schizophr. Res. 121, 125–130 (2010).
-
Iwata, Y. et al. Effects of glutamate positive modulators on cognitive deficits in schizophrenia: a systematic review and meta-analysis of double-blind randomized controlled trials. Mol. Psychiatry 20, 1151–1160 (2015).
-
Nomura, J. et al. Part for neonatal D-serine signaling: prevention of physiological and behavioral deficits in adult Pick1 knockout mice. Mol. Psychiatry 21, 386–393 (2016).
-
Marx, C. E. et al. Proof-of-concept randomized controlled trial of pregnenolone in schizophrenia. Psychopharmacology (Berl) 231, 3647–3662 (2014).
-
Vinkers, C. H., Mirza, N. R., Olivier, B. & Kahn, R. S. The inhibitory GABA system as a therapeutic target for cerebral symptoms in schizophrenia: investigational agents in the pipeline. Exp. Opin. Investig. Drugs xix, 1217–1233 (2010).
-
Rudolph, U. & Mohler, H. gabaa receptor subtypes: therapeutic potential in down syndrome, affective disorders, schizophrenia, and autism. Annu. Rev. Pharmacol. Toxicol. 54, 483–507 (2014).
-
Gill, M. M. & Grace, A. A. The role of alpha5 GABAA receptor agonists in the treatment of cerebral deficits in schizophrenia. Curr. Pharm. Des. twenty, 5069–5076 (2014).
-
Buchanan, R. W. et al. A randomized clinical trial of MK-0777 for the treatment of cerebral impairments in people with schizophrenia. Biol. Psychiatry 69, 442–449 (2011).
-
Oberman, L. M., Pascual-Leone, A. & Rotenberg, A. Modulation of corticospinal excitability by transcranial magnetic stimulation in children and adolescents with autism spectrum disorder. Front. Hum. Neurosci. 8, 627 (2014).
-
Wang, D. D. & Kriegstein, A. R. Blocking early GABA depolarization with bumetanide results in permanent alterations in cortical circuits and sensorimotor gating deficits. Cereb Cortex 21, 574–587 (2011).
-
Bruining, H. et al. Paradoxical benzodiazepine response: a rationale for bumetanide in neurodevelopmental disorders? Pediatrics 136, e539–e543 (2015).
-
Meck, W. H. & Williams, C. Fifty. Choline supplementation during prenatal development reduces proactive interference in spatial retentiveness. Brain Res. Dev. Brain Res. 118, 51–59 (1999).
-
Ross, R. G. et al. Perinatal choline effects on neonatal pathophysiology related to later on schizophrenia chance. Am. J. Psychiatry 170, 290–298 (2013).
-
Steullet, P. et al. Redox dysregulation, neuroinflammation, and NMDA receptor hypofunction: A "central hub" in schizophrenia pathophysiology? Schizophr. Res.; e-pub ahead of print (2014).
-
Miller, B. J., Culpepper, N. & Rapaport, M. H. C-reactive protein levels in schizophrenia: a review and meta-assay. Clin. Schizophr. Relat. Psychoses 7, 223–230 (2014).
-
Fineberg, A. M. & Ellman, 50. M. Inflammatory cytokines and neurological and neurocognitive alterations in the course of schizophrenia. Biol. Psychiatry 73, 951–966 (2013).
-
Coughlin, J. M. et al. Marked reduction of soluble superoxide dismutase-1 (SOD1) in cerebrospinal fluid of patients with recent-onset schizophrenia. Mol. Psychiatry 18, 10–11 (2013).
-
Bergink, V., Gibney, South. M. & Drexhage, H. A. Autoimmunity, inflammation, and psychosis: a search for peripheral markers. Biol. Psychiatry 75, 324–331 (2014).
-
Monin, A. et al. Glutathione deficit impairs myelin maturation: relevance for white matter integrity in schizophrenia patients. Mol. Psychiatry xx, 827–838 (2014).
-
Berk, M., Malhi, G. S., Gray, L. J. & Dean, O. M. The promise of Northward-acetylcysteine in neuropsychiatry. Trends Pharmacol. Sci. 34, 167–177 (2013).
-
Powell, S. B., Sejnowski, T. J. & Behrens, M. M. Behavioral and neurochemical consequences of cortical oxidative stress on parvalbumin-interneuron maturation in rodent models of schizophrenia. Neuropharmacology 62, 1322–1331 (2012).
-
Jiang, Z., Rompala, Yard. R., Zhang, S., Cowell, R. One thousand. & Nakazawa, M. Social isolation exacerbates schizophrenia-like phenotypes via oxidative stress in cortical interneurons. Biol. Psychiatry 73, 1024–1034 (2013).
-
Cabungcal, J. H. et al. Juvenile antioxidant treatment prevents developed deficits in a developmental model of schizophrenia. Neuron 83, 1073–1084 (2014).
-
Hashimoto, K. Targeting of NMDA receptors in new treatments for schizophrenia. Exp. Opin. Ther. Targets eighteen, 1049–1063 (2014).
-
Giordano, E. & Visioli, F. Long-chain omega 3 fat acids: molecular bases of potential antioxidant actions. Prostaglandins Leukot. Essent. Fatty Acids 90, ane–four (2014).
-
Lorente-Cebrian, S. et al. An update on the role of omega-3 fat acids on inflammatory and degenerative diseases. J. Physiol. Biochem. 71, 341–349 (2015).
-
Amminger, G. P., Schafer, M. R., Schlogelhofer, Yard., Klier, C. Yard. & McGorry, P. D. Longer-term outcome in the prevention of psychotic disorders past the Vienna omega-three written report. Nat. Commun. 6, 7934 (2015).
-
Markulev, C. et al. NEURAPRO-Eastward study protocol: a multicentre randomized controlled trial of omega-iii fatty acids and cognitive-behavioural case direction for patients at ultra loftier risk of schizophrenia and other psychotic disorders. Early Interv. Psychiatry; e-pub alee of print (2015).
-
Ryan, A. S. et al. Effects of long-chain polyunsaturated fat acrid supplementation on neurodevelopment in childhood: a review of human studies. Prostaglandins Leukot. Essent. Fatty Acids 82, 305–314 (2010).
-
Dalsgaard, South. et al. Association between Attention-Arrears Hyperactivity Disorder in babyhood and schizophrenia later in adulthood. Eur. Psychiatry 29, 259–263 (2014).
-
Maibing, C. F. et al. Take a chance of schizophrenia increases afterward all kid and adolescent psychiatric disorders: a nationwide written report. Schizophr. Bull. 41, 963–970 (2014).
-
Keightley, P. C., Koloski, Northward. A. & Talley, N. J. Pathways in gut-brain communication: show for distinct gut-to-brain and brain-to-gut syndromes. Aust. NZ J. Psychiatry 49, 207–214 (2015).
-
Smythies, L. E. & Smythies, J. R. Microbiota, the immune system, black moods and the encephalon-melancholia updated. Front. Hum. Neurosci. 8, 720 (2014).
-
Tomasik, J., Yolken, R. H., Bahn, S. & Dickerson, F. B. Immunomodulatory effects of probiotic supplementation in schizophrenia patients: a randomized, placebo-controlled trial. Biomark Insights 10, 47–54 (2015).
-
Mohammadi, A. A. et al. The furnishings of probiotics on mental health and hypothalamic-pituitary-adrenal centrality: a randomized, double-blind, placebo-controlled trial in petrochemical workers. Nutr. Neurosci.; east-pub ahead of impress (2015).
-
Aas, M., Dazzan, P. et al. Abnormal cortisol awakening response predicts worse cognitive function in patients with first-episode psychosis. Psychol. Med. 41, 463–476 (2011).
-
Hempel, R. J. et al. Diurnal cortisol patterns of young male patients with schizophrenia. Psychiatry Clin. Neurosci. 64, 548–554 (2010).
-
Walder, D. J., Walker, Eastward. F. & Lewine, R. J. Cognitive functioning, cortisol release, and symptom severity in patients with schizophrenia. Biol. Psychiatry 48, 1121–1132 (2000).
-
Pruessner, M. et al. Attenuated cortisol response to acute psychosocial stress in individuals at ultra-high adventure for psychosis. Schizophr. Res. 146, 79–86 (2013).
-
Walker, E. F. et al. Cortisol levels and risk for psychosis: initial findings from the Northward American prodrome longitudinal study. Biol. Psychiatry 74, 410–417 (2013).
-
Nordholm, D. et al. Pituitary gland volume in patients with schizophrenia, subjects at ultra high-chance of developing psychosis and healthy controls: a systematic review and meta-assay. Psychoneuroendocrinology 38, 2394–2404 (2013).
-
Garcia-Bueno, B., Caso, J. R. & Leza, J. C. Stress equally a neuroinflammatory condition in brain: damaging and protective mechanisms. Neurosci. Biobehav. Rev. 32, 1136–1151 (2008).
-
Vaessen, T., Hernaus, D., Myin-Germeys, I. & van Amelsvoort, T. The dopaminergic response to acute stress in health and psychopathology: a systematic review. Neurosci. Biobehav. Rev. 56, 241–251 (2015).
-
Schiavone, S., Jaquet, V., Trabace, 50. & Krause, K. H. Severe life stress and oxidative stress in the brain: from animal models to man pathology. Antioxid. Redox Signal. 18, 1475–1490 (2013).
-
Hildebrandt, T. & Greif, R. Stress and addiction. Psychoneuroendocrinology 38, 1923–1927 (2013).
-
Manetti, 50., Cavagnini, F., Martino, Due east. & Ambrogio, A. Effects of cocaine on the hypothalamic-pituitary-adrenal axis. J. Endocrinol. Invest. 37, 701–708 (2014).
-
Waasdorp, T. East., Bradshaw, C. P. & Leaf, P. J. The affect of schoolwide positive behavioral interventions and supports on bullying and peer rejection: a randomized controlled effectiveness trial. Arch. Pediatr. Adolesc. Med. 166, 149–156 (2012).
-
Cullen, A. E., Fisher, H. Fifty., Roberts, R. East., Pariante, C. M. & Laurens, Yard. R. Daily stressors and negative life events in children at elevated risk of developing schizophrenia. Br. J. Psychiatry 204, 354–360 (2014).
-
van Dam, D. Due south. et al. Childhood bullying and the association with psychosis in non-clinical and clinical samples: a review and meta-analysis. Psychol. Med. 42, 2463–2474 (2012).
-
Stoltz, S. et al. Effectiveness of an individual school-based intervention for children with aggressive behaviour: a randomized controlled trial. Behav. Cogn. Psychother. 41, 525–548 (2013).
-
Lereya, South. T., Copeland, Westward. E., Costello, Eastward. J. & Wolke, D. Adult mental health consequences of peer bullying and maltreatment in childhood: ii cohorts in ii countries. Lancet Psychiatry ii, 524–531 (2015).
-
Kelleher, I. et al. Childhood trauma and psychosis in a prospective cohort study: cause, effect, and directionality. Am. J. Psychiatry 170, 734–741 (2013).
-
Norberg, M. 1000., Kezelman, S. & Lim-Howe, N. Primary prevention of cannabis use: a systematic review of randomized controlled trials. PLoS ONE 8, e53187 (2013).
-
Patnode, C. D. et al. Primary care behavioral interventions to forbid or reduce illicit drug use and nonmedical pharmaceutical apply in children and adolescents: a systematic evidence review for the U.Due south. Preventive Services Chore Forcefulness. Ann. Intern. Med. 160, 612–620 (2014).
-
Vogl, L. E., Newton, North. C., Champion, G. E. & Teesson, G. A universal harm-minimisation approach to preventing psychostimulant and cannabis use in adolescents: a cluster randomised controlled trial. Subst. Abuse Treat. Prev. Policy 9, 24 (2014).
-
Deste, G. et al. Persistence of effectiveness of cognitive remediation interventions in schizophrenia: a ane-year follow-up study. Schizophr. Res. 161, 403–406 (2015).
-
Kontis, D., Huddy, Five., Reeder, C., Landau, South. & Wykes, T. Effects of age and cognitive reserve on cognitive remediation therapy outcome in patients with schizophrenia. Am. J. Geriatr. Psychiatry 21, 218–230 (2013).
-
Bowie, C. R., Grossman, M., Gupta, M., Oyewumi, 50. Yard. & Harvey, P. D. Cognitive remediation in schizophrenia: efficacy and effectiveness in patients with early versus long-term grade of illness. Early on Interv. Psychiatry eight, 32–38 (2014).
-
Vita, A. et al. Predictors of cognitive and functional improvement and normalization after cognitive remediation in patients with schizophrenia. Schizophr. Res. 150, 51–57 (2013).
-
Fisher, M., Loewy, R., Hardy, K., Schlosser, D. & Vinogradov, South. Cognitive interventions targeting brain plasticity in the prodromal and early on phases of schizophrenia. Annu. Rev. Clin. Psychol. 9, 435–463 (2013).
-
Cotman, C. W. & Berchtold, N. C. Do: a behavioral intervention to raise encephalon wellness and plasticity. Trends Neurosci. 25, 295–301 (2002).
-
Draganski, B. et al. Neuroplasticity: changes in grey matter induced by training. Nature 427, 311–312 (2004).
-
Douw, 50., Nieboer, D., van Dijk, B. Westward., Stam, C. J. & Twisk, J. W. A healthy brain in a good for you trunk: brain network correlates of concrete and mental fitness. PLoS ONE 9, e88202 (2014).
-
Lee, T. Yard. et al. Aerobic practice interacts with neurotrophic factors to predict cerebral functioning in adolescents. Psychoneuroendocrinology 39, 214–224 (2014).
-
Gottesman, I. I. & Shields, J. A polygenic theory of schizophrenia. Proc. Natl Acad. Sci. USA 58, 199–205 (1967).
-
Rasic, D., Hajek, T., Alda, K. & Uher, R. Take a chance of mental illness in offspring of parents with schizophrenia, bipolar disorder, and major depressive disorder: a meta-analysis of family unit loftier-risk studies. Schizophr. Bull. 40, 28–38 (2014).
-
Bassett, A. S. et al. The schizophrenia phenotype in 22q11 deletion syndrome. Am. J. Psychiatry 160, 1580–1586 (2003).
-
Duijff, S. North. et al. Cognitive evolution in children with 22q11.two deletion syndrome. Br. J. Psychiatry 200, 462–468 (2012).
-
Goodman, B. One thousand., Rutberg, J., Lin, W. W., Pulver, A. East. & Thomas, G. H. Hyperprolinaemia in patients with deletion (22)(q11.2) syndrome. J. Inherit. Metab. Dis. 23, 847–848 (2000).
-
Crump, F. T., Fremeau, R. T. & Craig, A. M. Localization of the brain-specific high-affinity l-proline transporter in cultured hippocampal neurons: molecular heterogeneity of synaptic terminals. Mol. Cell. Neurosci. 13, 25–39 (1999).
-
Cohen, S. G. & Nadler, J. V. Proline-induced potentiation of glutamate transmission. Brain Res. 761, 271–282 (1997).
-
Raux, G. et al. Involvement of hyperprolinemia in cognitive and psychiatric features of the 22q11 deletion syndrome. Hum. Mol. Genet. 16, 83–91 (2007).
-
Vorstman, J. A. et al. Proline affects brain part in 22q11DS children with the low activity COMT 158 allele. Neuropsychopharmacology 34, 739–746 (2009).
-
Rees, E. et al. Analysis of copy number variations at xv schizophrenia-associated loci. Br. J. Psychiatry. 204, 108–114 (2014).
-
Vorstman, J. A. & Ophoff, R. A. Genetic causes of developmental disorders. Curr. Opin. Neurol. 26, 128–136 (2013).
-
Laurens, K. R., Hobbs, M. J., Sunderland, M., Dark-green, M. J. & Mould, G. Fifty. Psychotic-like experiences in a community sample of 8000 children aged ix to 11 years: an particular response theory analysis. Psychol. Med. 42, 1495–1506 (2012).
-
Poulton, R. et al. Children's self-reported psychotic symptoms and developed schizophreniform disorder: a 15-year longitudinal study. Arch. Gen. Psychiatry 57, 1053–1058 (2000).
-
Fisher, H. L. et al. Specificity of babyhood psychotic symptoms for predicting schizophrenia by 38 years of age: a birth cohort report. Psychol. Med. 43, 2077–2086 (2013).
-
Mittal, V. A., Dean, D. J., Mittal, J. & Saks, E. R. Ethical, legal, and clinical considerations when disclosing a high-risk syndrome for psychosis. Bioethics 29, 543–556 (2015).
-
Appelbaum, P. S. Ethical challenges in the primary prevention of schizophrenia. Schizophr. Balderdash. 41, 773–775 (2015).
-
Fineberg, North. A. et al. The size, burden and cost of disorders of the brain in the UK. J. Psychopharmacol. 27, 761–770 (2013).
-
Agnew-Blais, J. C. et al. Early babyhood IQ trajectories in individuals later developing schizophrenia and affective psychoses in the new england family unit studies. Schizophr. Bull. 41, 817–823 (2015).
-
Cannon, T. D. et al. Progressive reduction in cortical thickness equally psychosis develops: a multisite longitudinal neuroimaging study of youth at elevated clinical take a chance. Biol. Psychiatry 77, 147–157 (2015).
-
Nieman, D. H. et al. Psychosis prediction: stratification of gamble estimation with information-processing and premorbid functioning variables. Schizophr. Bull. 40, 1482–1490 (2014).
-
Gale, C. R. et al. Cognitive ability in early adulthood and take a chance of 5 specific psychiatric disorders in eye age: the Vietnam experience study. Arch. Gen. Psychiatry 65, 1410–1418 (2008).
Author information
Affiliations
Contributions
I.Southward. and J.V. were involved in overall concept of paper and figures, literature search, and writing. C.E.B., S.N.D., K.Thou., T.vA, L.dH., C.H.Five., R.E.1000., C.A., and C.1000.D.-C. were involved in writing. E.vD. was involved in writing and design of Figure ane. E.J.B. was involved in writing and co-design of Figure 2.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This piece of work is licensed under a Creative Eatables Attribution iv.0 International License. The images or other third party textile in this article are included in the article'southward Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a re-create of this license, visit http://creativecommons.org/licenses/by/iv.0/
Reprints and Permissions
Nigh this commodity
Cite this commodity
Sommer, I., Bearden, C., van Dellen, E. et al. Early interventions in take a chance groups for schizophrenia: what are we waiting for?. npj Schizophr 2, 16003 (2016). https://doi.org/10.1038/npjschz.2016.3
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/npjschz.2016.three
Farther reading
Source: https://www.nature.com/articles/npjschz20163
0 Response to "Rate of Occurrence Schizophrenia Study Color and Babies"
Post a Comment